Product Description
The gold nanoparticles or colloidal gold offered by TORSKAL are produced by green chemistry (by bio-reduction of metal salts HAuCl4), with crude and / or purified extracts from plants. These extracts have the double action of reducing the metal and stabilizing the formed nanoparticles. The synthesis of nanoparticles by using the bio-reducing potential of plant extracts has been demonstrated in various studies in recent years. Based on this observation, TORSKAL developed a green chemistry approach to synthesize nanoparticles using endemic plants from Reunion Island.
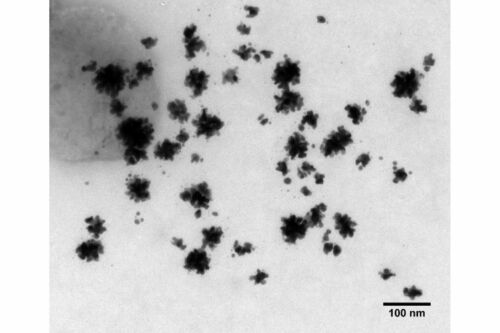
Each batch of gold nanoparticles or colloidal gold are fully characterized by transmission electron microscopy (TEM), dynamic light scattering (DLS), zeta-potential, XRD, UV-Vis, FTIR and Raman spectroscopy to understand their physicochemical and biological properties.

TORSKAL’s AuNPs synthesized from green chemistry, lyophilised plant extract, plant powder & dried plant leaves
Applications of our gold nanoparticles:
Product Specification
| Parameters | OD : 1 | OD : 10 | OD : 50 |
|---|---|---|---|
| Diameter (TEM) (nm) | 14.83 | Same | Same |
| Size disparity (+/- nm) | 2.84 | Same | Same |
| Diameter deviation | 15% | Same | Same |
| SPR Peak | 537 – 540 | Same | Same |
| Particle concentration (NP/mL) | 1.27E + 12 | 1.27E + 13 | 6.35E + 13 |
| Mass concentration (mg/mL) | 4.17E – 2 | 4.17E – 1 | 2.09E + 00 |
| Particle molar concentration | 2.19E – 9 | 2.19E – 8 | 1.05E – 07 |
| Zeta potential (mV) | -26.2 | Same | Same |
| Zeta potential deviation | 10% | Same | Same |
| Particle volume (nm3) | 1.71E + 3 | Same | Same |
| Particle surface (nm2) | 6.31E + 02 | Same | Same |
| Surface/Volume Ratio | 0.404 | Same | Same |
| Solvent | Water | Same | Same |
| Stability | 3 months | Same | Same |
| Storage temperature | 4 – 8 °C | Same | Same |
| Color | Purple-red | Same | Same |
Physico-chemical Characterization
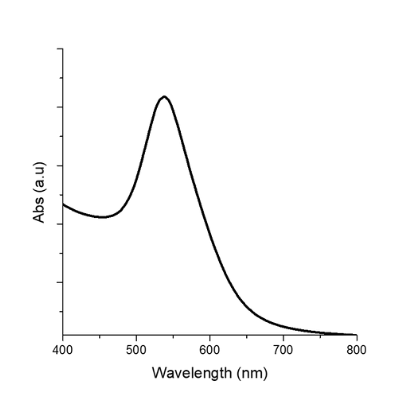
Extinction spectrum of the solution of SAuNPs 15
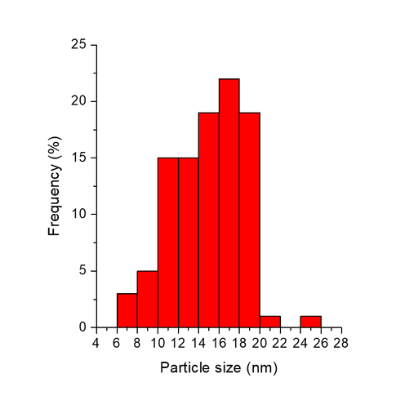
Size distribution of SAuNPs 15 determined by transmission electron microscopy image
The unique properties of TORSKAL’s green gold nanoparticles:
Conformity
We employ an environmentally friendly technique for the production of well-characterized gold nanoparticles.
Salt resistance
Stable over 0.4 M NaCl, which is 5 times higher than gold nanoparticles synthesized by the citrate reducing method.
Scalability
Our process of producing gold nanoparticles or colloidal gold is very straightforward to scale up with a lower risk of contamination.
Biocompatibility
Our green synthesis process does not require toxic chemicals, which makes our products suitable for biomedical applications.
High purity
Our gold nanoparticles are of high purity with the absence of aggregation, which makes them monodisperse gold nanoparticles.
High stability – For at least 3 months
Our gold nanoparticles are more stable than Turkevich gold nanoparticles in a saline environment, over time, and after centrifugation.